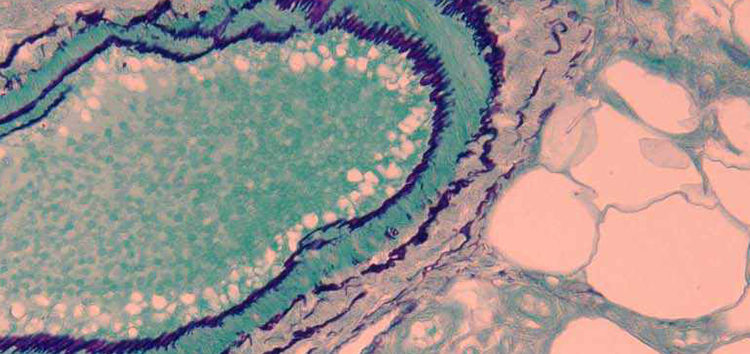
Aldehyde fuchsin stains certain nonionic proteins and peptides in unoxidized cells and tissues. These substances include granules of pancreatic islet B cells, elastic fibers and hepatitis B surface antigen.
Saffron Scientific Histology Services
Histology Services

Aldehyde fuchsin stains certain nonionic proteins and peptides in unoxidized cells and tissues. These substances include granules of pancreatic islet B cells, elastic fibers and hepatitis B surface antigen.
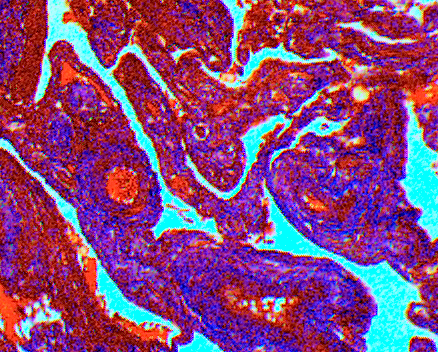
The purpose of this stain is to differentiate between collagen & muscle. Muscle should stain red with collagen staining blue. Let’s take a look at the staining components and how they affect the stain. Two things that affect trichrome staining are fixation and differentiation.
First and foremost, fixation plays an important role in this stain. Bouin’s solution is the preferred fixative. Tissues fixed in NBF required post fixation in Bouin’s for proper staining. The acid pH is necessary to increase the selectivity of aniline blue for the collagen fibers. Recommended times for post fixing in Bouin’s vary from 1 hour at 56°C or overnight at room temperature. Please note that Bouin’s contains picric acid that is explosive when dry. Heating solutions that contain picric acid increases the potential of drying picric acid. Alternative suggested post fixation can be achieved with Gram’s iodine and citrate buffer.
Weigert’s iron hematoxylin is recommended for nuclear staining in trichromes. It is prepared fresh by mixing equal proportions of stock solution A, alcoholic hematoxylin, with solution B, acidified ferric chloride. Increasing the concentrations of Weigert’s iron hematoxylin stock solutions A from 1% to 2% hematoxylin and stock solution B from 29% to 62% will result in a darker nuclear stain.
The Biebrich scarlet-acid fuchsin stain mixture binds to all tissue components because of its small size. This includes both muscle & collagen. The phosphotungstic and phosphomlybdic acids selectively remove these dyes from collagen and also act as a mordant for the larger dye molecules of aniline blue. If the biebrich scarlet-acid fuchsin is insufficiently removed from the collagen the aniline blue stains poorly or not at all. Phospho acids work best when freshly prepared. If storage is necessary, refrigerate solutions. Phosphotungstic is unstable at a pH greater than 2.
Fading of Aniline blue staining of collagen can be caused by over differentiation of slides in 1% glacial acetic acid. Decreasing time and/or concentration in acetic acid may prevent fading.
References:
Luna LG (Ed.): Manual of Histologic Staining Methods of the Armed Forces Institute of Pathology, 3rd Edition. McGraw-Hill, New York, 1968
Yufeng Y, Chapman C. (2013) Masson Trichrome Stain: Postfixation Substitutes, Journal of Histotechnology, 26:2, 131-134,
Cheryl H. Crowder (1983) The Trichrome Stain: A Modification, Journal of Histotechnology, 6:3, 133-134,

Histology Laboratory Ergonomics
Ergonomics is the science of designing the job, equipment, and workplace to fit the worker. Why should the Histotechnologist be concerned about laboratory ergonomics? As Histotechnologists we perform daily tasks that are precise, repetitive, and in a fast-paced manner. Work-related musculoskeletal disorders currently account for one-third of all occupational injuries and illnesses reported to the Bureau of Labor Statistics (BLS) by employers every year. When a movement is done repeatedly such as while using a manual microtome or the “pinch grip” used with forceps during the embedding process, a stress injury can form. These small injuries build up over time and can develop into Musculoskeletal Disorders (MSD). Most frequently these injuries affect the back, neck, shoulders, and upper limbs; less frequently the lower limbs. There are ways to improve the ergonomics of your workspace without spending too much money. This discussion will be focused on arm related issues in the hand and shoulder.
Ergonomic Risk Area- Manual Microtome


Ergonomic Risk Area- Forceps Pinch Grip

-Ergonomic design has the widest gripping area balanced in the center of the tool

-Standard design
While not everyone can afford to purchase specially designed ergonomic tools there are modifications that can be made to existing tools.

This photo from the UC Berkley Tang Center shows a great way to improve the “pinch grip” of forceps by using inexpensive plastic-coated foam tubing. This tubing can be found in many hobby or craft stores and it comes in a multitude of sizes. By using either specially designed tools or simple modifications to existing tools the ergonomics of your workplace can be improved and the risk of injury reduced.
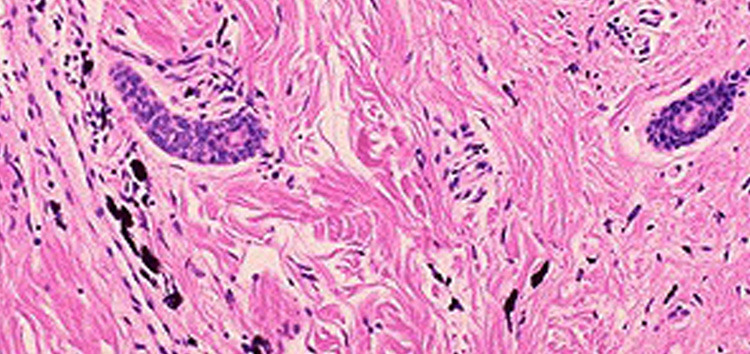
Saffron Scientific Histology Services provides high-quality H&E’s for research and diagnostic interpretation.
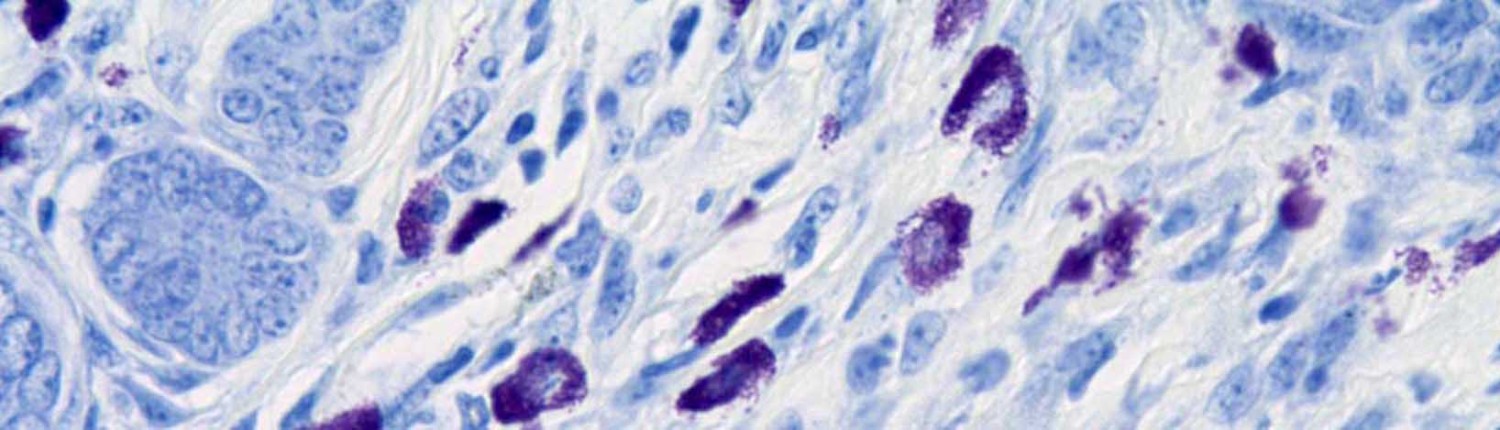
Saffron Scientific Histology Services offers a variety of special stains for paraffin sections.
